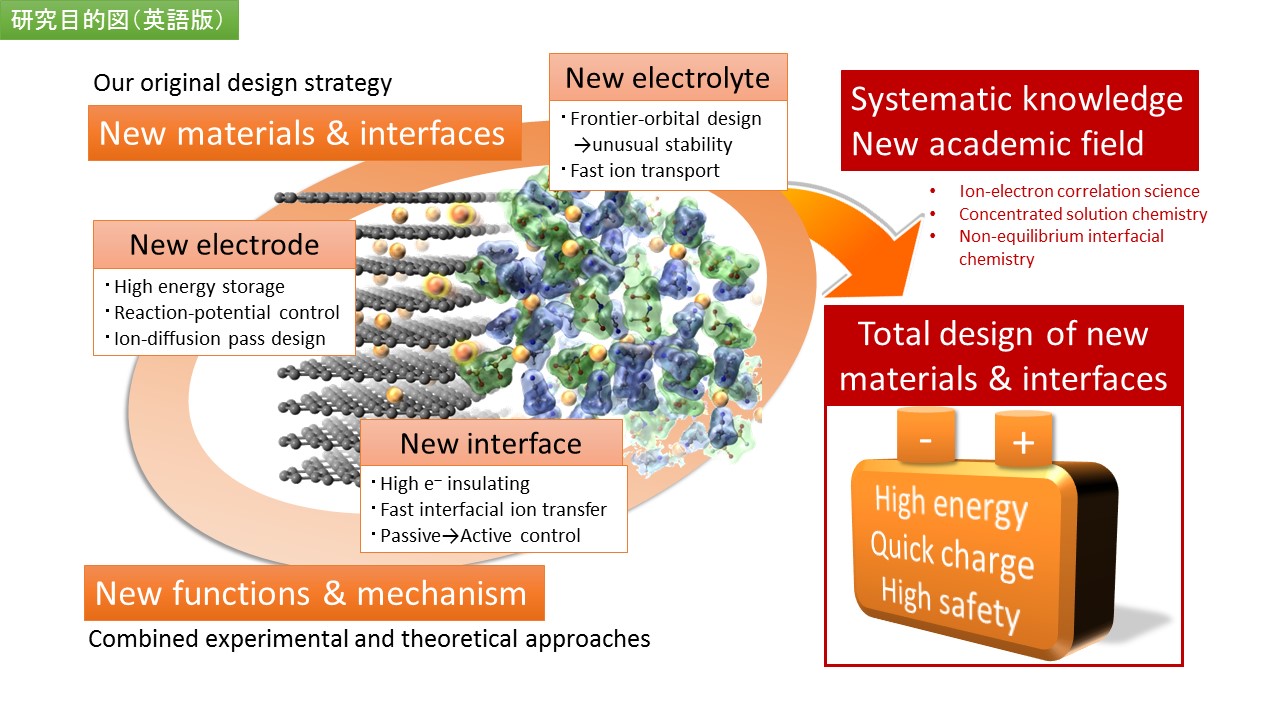
Research
Electrode materials
The rechargeable batteries are recognized as a powerful solution toward sustainable society. In order to put this promising technology into realistic solution with much more advanced batteries, we have continuously identified wide variety of new electrode(cathode/anode) materials with our original exploration strategies. These commitments are not based on just the intuitive idea but on our accumulated knowledge/output and original directions, and we have indeed discovered so many new materials so far.
Electrolyte
Lithium-ion battery is the most advanced chemical energy storage system but its application is mostly limited to portable electronics, due to its limited capacity and high cost. Major reasons are in limited materials properties and hence limited number of practical materials; only a few electrode materials can be applied to the practical batteries. This situation has forced to apply only one-kind of ethylene carbonate-based electrolyte for more than 20 years. One of our original core-competence is super concentrated organic electrolytes, which we have proved to be able to largely expand the possible combination of salts and solvents.
Theoretical approach
Atomic-scale comprehension of the reaction mechanism is critical for development of superior batteries. We have applied the ab-initio methods to investigate the structure, electronic state, ionic diffusion, ion adsorption, solvation, and interphase. Particularly, we are to predict the character of electrode/electrolyte interface. Comprehensive scientific understanding of the reaction will lead to dramatic improvement of energy density and power density of batteries.
Spectroscopic approach
The state-of-the-art spectroscopic methods using the synchrotron source enable us to probe/monitor/visualize the structure and electronic state at bulk and surface of the electrodes/electrolytes/interfaces. We have applied the various diffraction, absorption, and resonant techniques, providing a platform for implementing the multi-scale understanding of the electrode reaction.